
Excellent Outcomes with New Minimally Invasive Heart Surgeries
The growth of transcatheter aortic valve replacements (TAVRs) and MitraClips has been “revolutionary,” says Satya Shreenivas, MD, assistant professor and director of the structural heart disease program at the University of Cincinnati Heart, Lung and Vascular Institute. The Institute’s focus is implanting heart valves nonsurgically by transcatheter aortic valve replacement as well as nonsurgical repair of heart valves.
The use of TAVRs began in the early 2000s and resulted in lower risk of infection, reduced trauma to chest and heart muscle, fewer hospital days, and shortened recovery time.1 The iterations in the devices have produced smaller, improved versions, resulting in less leakage and fewer postsurgical complications, such as bleeding and stroke, Dr. Shreenivas says. These are the biggest advantages in the current treatment of transcatheter aortic valves. According to a review of two long-term clinical trials, Placement of Aortic Transcatheter Valves (PARTNER) I and CoreValve US Pivotal trials, and national registries, use of TAVRs has showed long-term effectiveness and reliability, although there appear to be outcome differences with valve types.2 Benefits of minimally invasive treatment options for heart short-term efficacy and safety evaluations of newer, improved versions also have shown good results.2 Further, their use is no longer aimed only at high-risk patients; similarly good outcomes have been demonstrated in intermediate-risk patients.2 Studies of cost-effectiveness, however, indicate greater total costs associated with TAVR, especially for intermediate-risk patients.3
Current treatments for mitral valve regurgitation are medication, open heart surgery, and minimally-invasively using the transcatheter MitraClip mitral valve repair system (manufactured by Abbott). Dr. Shreenivas described MitraClip, a permanent implant that clips together the sides of the mitral valves, as a very good treatment for certain patients with specific types of leaks (i.e., not for patients with a damaged heart wall or leaks in areas other than the central part of the valve). Studies have evaluated the effectiveness of MitraClip therapy in patients with refractory heart failure and mitral regurgitation. In 45 patients with both conditions, use of MitraClip was found to have sustained clinical benefit and reduced the rate of heart failure re-hospitalization.4
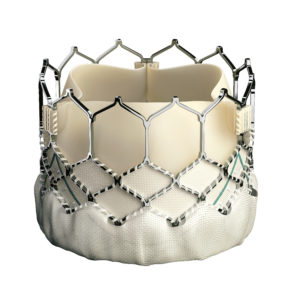
Edwards SAPIEN 3 Transcatheter Heart Valve. Image courtesy of Edwards Lifesciences LLC, Irvine, CA. Edwards, Edwards Lifesciences, Edwards SAPIEN, SAPIEN, SAPIEN XT and SAPIEN 3 are trademarks of Edwards Lifesciences Corporation.
In a Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy (STS/ACC TVT) Registry study (i.e., all 7,710 eligible United States TAVR cases) from 224 participating registry hospitals, 92% of cases achieved successful device implantation, with an in-hospital mortality rate of 5.5%, and a stroke rate of 2.0%.5 An analysis of sex-specific outcomes in patients in the PARTNER trial found that women were more likely than men to be alive one year after TAVR, although they experienced more major bleeding and vascular complications at 30 days.6
Improvements to MitraClip are on the horizon, and they will transform the treatment of mitral valve disease, says Dr. Shreenivas. Edwards Lifesciences Corp., which manufactures TAVRs and mitral valves, has received approval from the U.S. Food and Drug Administration for an investigational device exemption to conduct an early feasibility study, with a goal of enrolling 20 patients to test its mitral valve device.7 Dr. Shreenivas looks forward to University of Cincinnati Medical Center’s participation in larger trials that will follow this preliminary study. He also expects that Medtronic, a medical device company, will test a transcatheter mitral valve.
According to Dr. Shreenivas, clinical gaps “absolutely” remain in the population with valve disease that neither therapy addresses. There are different types and severities of valve disease, not all of which will have optimal benefit from surgery. Clinicians are considering minimally invasive surgery in younger patients with aortic stenosis (i.e., people ranging from age 50 to 70) when aortic and mitral valve surgery could have long-lasting benefits, beginning at a younger age. Edwards Lifesciences has announced a low-risk trial for these patients, and if the trial results (expected in approximately two years) are positive, the treatment of choice for aortic stenosis will be transcatheter surgery, not valve replacement, says Dr. Shreenivas.
UC Medical Center’s minimally-invasive heart surgery program includes a core team of a surgeon, interventionalists, an anesthesiologist, and an echocardiologist, plus catheterization lab nurses, technicians, and operating room staff. Surgeries are performed in a new hybrid operating room that offers advanced surgical facilities and imaging devices. So far, patient results have been excellent, and Dr. Shreenivas looks forward to seeing similar outcomes with improving techniques.
References
- Goldstone AB, Joseph Woo Y. Minimally invasive surgical treatment of valvular heart disease. Semin Thorac Cardiovasc Surg. 2014;26:36-43.
- Tsai MT, Tang GH, Cohen GN. Year in review: transcatheter aortic valve replacement. Curr Opin Cardiol. 2016;31:139-147.
- Ailawadi G, La Par DJ, Speir AM, et al. Contemporary costs associated with transcatheter aortic valve replacement: a propensity-matched cost analysis. Ann Thorac Surg. 2016;101:154-161.
- Adamo M, Barbanti M, Curetto S, et al. Effectiveness of MitraClip therapy in patients with refractory heart failure. J Interv Cardiol. 2015;28(1):61-68.
- Mack MJ, Brennan JM, Brindis R, et al, for the STS/ACC TVT Registry. Outcomes following the transcatheter aortic valve replacement in the United States. JAMA. 2013;310(19):2069-2077.
- Kodali, S, Williams MR, Doshi D, et al. Sex-specific differences at presentation and outcomes among patients undergoing transcatheter aortic valve replacement: A cohort study. Ann Intern Med 2016: 377-384.
- Early feasibility study of the CardiAQ™ TMVI System (transfemoral and transapical DS). Available at: https://clinicaltrials.gov/ct2/show/NCT02515539?term=CardiAQ&rank=1. Accessed March 7, 2016.
 Satya S. Shreenivas, MD
Satya S. Shreenivas, MD
Assistant Professor of Clinical Medicine
Department of Internal Medicine
Division of Cardiovascular Health and Disease
Director, Center for Structural and Adult Congenital Heart Disease
University of Cincinnati Heart, Lung and Vascular Institute
(513) 558-3344
shreensa@ucmail.uc.edu


